About Us
- TOP
- About Us
For Progress in Cancer Treatment
The Japan Clinical Oncology Group (JCOG) is the largest Japanese cooperative group, and is funded primarily by the National Cancer Center Research and Development Fund. JCOG's primary mission is to establish and advance standard treatments for various types of cancers. In order to achieve this mission, we are conducting a variety of multi-center, multi-disciplinary phase II or phase III trials that combine various modalities such as drugs, surgery, radiotherapy, and endoscopy. As of 2022, JCOG has approximately 50 trials open to accrual and 40 trials on follow-up.
JCOG is composed of study groups, committees, and a Data Center, Operations Office and Biorepository, all of which cooperate with each other in research activities.
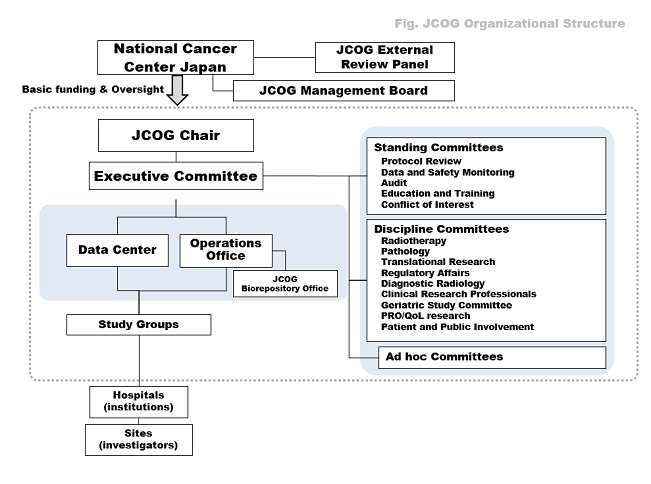
Study Groups
JCOG currently has 16 study groups covering all adult solid cancers, with each study group having its own participating institutions. In total, JCOG has about 190 active participating institutions all over Japan, with over 660 clinical sites. In order to perform multi-center clinical trials safely and smoothly, JCOG runs study group meetings and educational seminars for the medical professionals who participate in JCOG trials.
Committee
Several committees assess whether clinical trials are safely performed in accordance with the clinical study protocols. All JCOG study protocols are reviewed and approved by the Protocol Review Committee. All serious adverse events are submitted and reviewed by the Data and Safety Monitoring Committee (DSMC). The DSMC also regularly conducts in-person meetings for interim analyses. In order to assure high study quality, site visits and audits are performed by the JCOG Operations Office and the Audit Committee. The Radiotherapy Committee and Pathology Committee conduct quality control by means of central reviews. Other committees also play important roles in maintaining JCOG study quality.
Data Center/Operations Office
Responsible for JCOG's core functions, the Data Center and Operations Office conduct patient registration, data management, data analysis, protocol development, site visits and audits, investigator roster management, and so on. The JCOG Data Center and Operations Office are located in the National Cancer Center, Tokyo.
